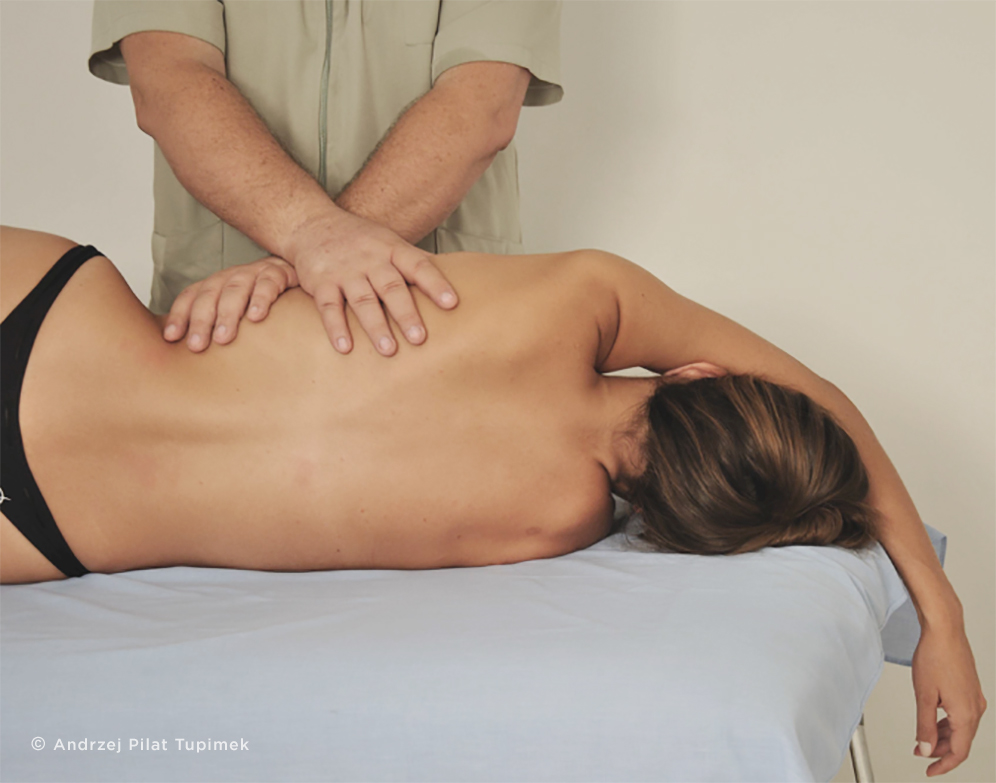
Definition of (MIT)
MIT is a therapeutic concept belonging to Manual Therapy aimed to the functional restoration of the altered fascial system. MIT is a process of assessment and treatment procedures in which the therapist transfers a slight force (traction and/or compression) to the target tissue (Pilat, 2012; Pilat, 2019), facilitating the recovery of the quality of the fascial system. The term "induction" is related to the facilitation of movement rather than a passive stretching of the fascial system. The result is a reciprocal reaction of the body that involves the biochemical and metabolic reaction, and finally, the physiological responses. This process aims to reshape the quality of the extracellular matrix of the connective tissue to facilitate and optimize the transfer of information to and within the fascial system (Chiquet, 2003; Wheeler, 2004). It is a process controlled by the central nervous system, in which the therapist acts as a facilitator (Pilat, 2019). The therapeutic action is concentrated in the provision of resources for the adjustment of the homeostatic balance. The ultimate goal is not the establishment of stable hierarchies but facilitating an optimal adaptation to the demands of the environment (Pilat, 2014). The result (change in body image, improvements in functional skills) should be evaluated and assessed not only by the therapist but also the patient. Myofascial induction aims to be a treatment that focuses on the patient (Pilat, 2015).
Definition of Fascia and characteristics of the Fascial System
There are different opinions about what we can define as fascia (Langevin, 20016, Langevin & Huijing, 2009, Schleip, Jäger & Kinler, 2012, Swanson, 2013, Stecco, 2014). The use of the expression fascial system is suggested. This system brings together different types of cells with different activities (in a similar way as, for example, the cardiovascular system or nervous system), and is related to other body systems through an uninterrupted and innervated structure of functional stability formed by the three-dimensional collagenous matrix (Kumka & Bonar, 2012, Pilat, 2014). This approach makes it easier to relate scientific and clinical findings and in turn offers a different and broader perspective for the analysis of body mechanics and pathomechanics. The aforementioned system represents a complex communication architecture (Kapandji, 2012) which ensures extensive mechano-receptive information, not only through its topographic distribution, but mainly due to the patterns of interrelation with other body structures (Lancerotto et al. 2011), especially the muscles. From its dynamic and fibrous construction, is distinguished the property of continuous remodeling (fascial plasticity) (Langevin, 2011), of aligning and adapting to the intrinsic and extrinsic tension applications of the body (Swanson, 2013). Tension alterations, created outside the physiological patterns of movement, can reorient body dynamics establishing retractable changes of the matrix (through the dynamics of myofibroblasts) (Tomasek, 2002), which affect freedom of movement (Gabbiani, 2007). The density, distribution and organoleptic characteristics of the fascial system differ in its route through the organism allowing it to adapt and respond to the movement's requests (Benjamin, 1995), however, its continuity is fundamental, which allows it to act as a synergistic whole, absorbing and distributing a local stimulus to the remaining elements of the set at different scales of its construction (Ingber, 2008). The intrinsic structural synergy of the fascial system assures the body the relative independence of the gravitational force, as well as enjoying a great capacity for adaptation, according to the requirements that come from outside and inside or, in relation to the availability of energy and nutrients in the environment (Nakajima et al. 2004). In addition to its structural function, fascia assumes and distributes the stimuli that the body receives: its network of receptors registers thermal, chemical, pressure, vibration and movement impulses; sending them to the homeostatic region of the central nervous system interoceptively (Craig, 2003). This way, an information potential linked by the system is created for a specific purpose (Pilat, 2012, Pilat, 2014).
Myofascial transmission of muscle contraction
The ubiquitous muscle contraction model based on the slippage of actin and myosin filaments described more than 40 years ago by Huxley & Simmons (1971), has supported the analysis of Newtonian body movement, characterized by the phenomenon of levers. In this model, the myofibrils organized in series, are independent motors that manage to approximate the myotendinous or myoaponeurotic extremes, thus triggering the movement. However, the discovery of the ultrastructure and the mechanobiology of the sarcomeral unit, have shaped a new model of myofibrils “embedded” within an extracellular matrix (MEC) that participates, from its own dynamics, in the contractile phenomenon (Gillies, 2011). The shortening of the myofibril exerts “multidirectional” forces within a fascial network (endomysium, perimisium and epimisium) organized under the tensegrity principles (Gillies, 2011, Purslow, 2010, Hujing, 2007). Most of these forces are destined to the myotendinous junction, however, approximately 30% use “epimisial” lateral transmission pathways, as is demonstrated in cases such as the aponeurotic expansion of the brachial biceps over the forearm (Eames et al. 2007) or the continuity of the pectoral fascia with the brachial (Stecco, 2008), that is, parallel to the tendinous pathways (Chi-Zhang, Gao, 2012, Huijing, 2007, Yaman A, 2013). The clinical importance of this phenomenon is exposed in the tendon transfers, where it is observed, that it is the extra-tendon intermuscular (epimisial) connections that govern the function on the extensor apparatus (Scott, 2003).
Innervation and proprioception of fascial system
Fascia is considered a neurosensitive structure that forms a complex functional network of interconnection and integration of body dynamics (Stecco et al. 2007). During muscle contraction, fascial expansions could transmit the contractile impulse to specific areas of the fascial system, stimulating proprioceptors in that area. The presence of the mechanoreceptors suggests an active participation of the fascia in proprioception, transmission of force and motor control. The proprioceptive role of the fascial network means that it can update the central nervous system, in terms of mechanical tension, to operate on the motor units at time, pace and level of adequate force. As a result, fascia is involved in the proprioceptive role (Van der Wal, 2009) and is vital for the proper functioning of the system (Basmajian & De Luca, 1985). Of interest is the network of free nerve endings type III and IV, also called interstitial mechanoreceptors, who are potential informants of stimuli of low and high mechanosensitive level.
For example, Group III afferent receptors are found in the perimuscular fascia and adventitia of muscular blood vessels and respond to a wide variety of stimuli, including pressure and stretching linked to matrix deformation after mechanical impulse application, this being applicable to the manual impulse used during the therapeutic process (Yi-Wen, 2009). Stimulation of afferent muscle receptors of group III and IV shows the presence of an important reflex inhibitory effect on alpha motor neurons and an excitation effect on gamma motor neuron (Kaufman, 2002). Dysfunction and pain related to the pathomechanical processes of the musculoskeletal system, suggests the presence of important anatomical and neurophysiological changes that also involve the fascial system (Alix, 1999). Pain and hypersensitivity can be caused by the activation and/or sensitization of peripheral nociceptors by endogenous substances (Mense, 2011). The presence of free (sensory) nerve determinations within the collagenous matrix of non-specialized connective tissue, mainly A δ and / or C fibers (Corey, 2011) reveals the possibility that fascial tissue is an important element of painful response. Studies on the innervation of the human thoracolumbar fascia (FTL) revealed its substantial innervation (Taguchi, Hoheisel & Mense, 2009). Von Tesarz et al. (2011) discovered that most of the nerve fibers are found in the superficial level of the FTL and in the superficial fascia, while the nerve fibers in the intermediate level are scarce. In parallel, nerve fibers that accompany blood vessels have been found in the superficial fascia. The location of most of the fibers around the blood vessels suggests that at least part of them are vasomotor fibers that, when activated, could cause ischemic pain (von Tesarz et al. 2011).
Miofascial Disfunction definition
The dysfunction of the Myofascial system is defined as an alteration of highly organized wave of specialized movements and the consequent inadequate transmission of information through the extracellular matrix (Pilat, 2003). The dysfunctional patterns of movement could facilitate variations in the mechanotransduction response (conversion of the mechanical impulse into the chemical response), with the consequent initiation of the molecular mechanisms that trigger the disease (Ingber, 2003).
The application of MIT is recommended mainly for patients with orthopedic, neuro-orthopedic, post-traumatic and degenerative dysfunctions related to the myofascial system.
General recommendations:
nervous system dysfunctions (central and peripheral)
pelvic floor disorders
circulatory system dysfunctions
TMJ dysfunction
sports injuries
respiratory disorders
Specific objectives:
mobilize superficial fascial restrictions
change the “stationary attitude” of collagen structures
facilitate the recovery of slip properties of the extracellular matrix
stimulate physiological orientation in fibroblast mechanics
prevent the formation of tissue adhesions
acquire a more efficient circulation of antibodies in the matrix
improve blood supply (histamine release)
improve blood supply to the nervous system
Bases of clinical applications (Pilat, 2003, Pilat, 2009, Pilat, 2014, Pilat, 2015)
In MIT, the practitioner applies a low intensity manual impulse to the fascial system. A tension impulse (traction and/or compression) prolonged over time is used (Barnes, 1990). Through it, the recovery of the quality of the fascial tissue (extracellular matrix) is facilitated (Martínez Rodríguez, Galán-del-Río, 2013). This process is mediated by molecular mechanisms associated with cellular mechanotransduction, piezoelectricity (Ahn, Grodzinsky, 2009), viscoelasticity (Chaudhry et al. 2007) and regulated "in real time" by the SNC (Craig, 2003, Urresti-López, 2011). It is mainly an instructive process, in the search for a new homeostatic level through the recovery of the range of motion, adequate tension, strength and ordination. The final result is transmitted in a better functionality with a lower energy expenditure.
Research related to some pathologies
An objective way to evaluate the effects of myofascial induction application techniques on muscle lesions with dynamic sonoelastography was reported by Martínez and Galán-del-Río (2013).
Leonard et al. (2009) reported that connective tissue manipulation improved peripheral circulation as also the healing processes in 20 patients with diabetic foot ulcers.
Significant differences between pre- and post-pressure measurements (pain thresholds), with decreased sensitivity at myofascial “trigger” points, were reported in several affected muscles: the long adductor (Roba & Pajaczkowski, 2009), the upper trapezium (Fryer & Hodgson, 2005) and cervical muscles (Hou et al. 2002).
Marshall et al. (2009) concluded that myofascial release helped reduce the severity and intensity of muscle pain in people with chronic fatigue syndrome.
Hicks (2009) reported that human fibroblasts secrete soluble mediators of myoblast differentiation and that myofascial release can regulate muscle development.
Tozzi et al. (2012) investigated non-specific lumbar pain, in relation to renal mobility. Using real-time ultrasound, this study demonstrated that "fascial osteopathic manipulation reduced the perception of pain and improved renal mobility."
Useros and Hernando (2008) concluded that myofascial induction has beneficial effects for patients with brain damage, with an emphasis on automatic posture control.
In patients with unilateral spatial neglect (an alteration of the position of the head with respect to the midline of the body), Vaquero Rodríguez (2012) observed significant results for the sensitivity variable in the experimental group treated with myofascial induction techniques, compared to those treated with Bobath therapy.
Fernández-Lao et al. (2011) applied myofascial release techniques in breast cancer survivors. The authors noted that myofascial release led to an immediate increase in the salivary flow rate, suggesting a parasympathetic effect of the intervention.
Vásquez et al. (2011) reported the efficacy of myofascial induction techniques in the treatment of "swimmer's shoulder" with respect to the balance of joint movement and pain.
Arguisuelas-Martínez (2010) demonstrated the effects of manipulation of the lumbar spine and thoracic-lumbar myofascial induction techniques in the activation pattern of trunk extensors.
In a double-blind study, Urresti-López (2011) applied the suboccipital induction technique in 26 subjects with chronic neck pain. Changes in the electroencephalogram (EEG) related to the reduction of latency in the experimental group were observed, compared to the control group. This result suggests that improvements in cognitive processes, including attention, memory and associative activation states are associated with the P300 wave. The absence of variants in other EEG parameters ruled out the influence of vascular modifications.
Heredia-Rizo et al. (2013) demonstrated that the application of the suboccipital muscle inhibition technique instantly improved the protruding position of the head. Additionally, the mechano-sensitivity of the major occipital nerve decreased immediately.
Ajimsha et al. (2012) presented an evidence, that myofascial release is more effective than control interventions to treat lateral epicondylitis, in computer professionals.
Castro-Sánchez et al. (2011a) in the study conducted in 74 patients with fibromyalgia (analysis of experimental and placebo groups), establish that myofascial release can improve pain, anxiety, sleep quality, depression and quality of life in patients with fibromyalgia.
Tozzi et al. (2012) concluded that myofascial release techniques are effective in releasing areas with reduced fascial mobility (slippage), as well as being effective in improving the perception of short-term pain in people with non-specific NP or low back pain.
Kuhar et al. (2007) reported a significant reduction in pain and an improvement in foot function as a short-term effect of myofascial release treatment.
Clinical investigations in healthy subjects
Fernández-Pérez et al. (2013) observed important immunological modulations, with an increase in B lymphocyte count after 20 minutes of the application of myofascial induction techniques in the craniocervical segment.
Toro et al. (2009) reported that the application of a single manual therapy session (including myofascial induction techniques) produced an immediate increase in heart rate variability and a decrease in tension, anger and perceived pain, in patients with headache problems related to chronic tension.
In a simple blind study, with the placebo group, Arroyo Morales et al. (2009) reported that myofascial induction techniques could encourage the recovery of a state of transient immunosuppression, induced by exercise in healthy and active women.
Arroyo Morales et al. (2008a) reported that heart rate variability and blood pressure recovery, after a physically stressful situation, were observed as a result of myofascial release, compared to false electrotherapy treatment.
Arroyo Morales et al. (2008b) reported that the application of an active recovery protocol, (application of global myofascial techniques), reduces the amplitude of EMG and vigor, when applied as a passive recovery technique, after high intensity exercise.
Henley et al. (2008) quantitatively demonstrated that cervical myofascial release modifies the sympatho-vagal balance, from the sympathetic to the parasympathetic nervous system.
In a study with 41 healthy young adults, randomized to the experimental and control groups, Fernández et al (2008) reported a significant decrease in anxiety levels after the application of myofascial induction treatment. In addition, significantly lower values of systolic blood pressure were observed, compared to baseline levels.
Saiz-LLamosas al. (2009) demonstrated the changes in cervical mobility ranges by performing Myofascial Induction of the nuchal ligament.